12. January 2015
Crystallising is a topic made for research and experiments. During the first couple of lessons crystallisation was the most popular subject. Hence we had loads of ideas to begin with. Since crystallisation is a very slow process it is important to start experimenting quickly, find a good method and eliminate all badly working materials.

We started off collecting ideas and began our first experiment trying to make rock candy. However, this apparently easy task turned out to be a failure, we eventually took it out after few days with no visible crystals being formed. We then tried a similar experiment with salt and soon saw our first results when the strings slowly started to harden and built up small crystals. This led us to our first idea for a possible project.
In the evening of our first day we built a cube out of plexiglass and wrapped strings around it and on the inside. We proceeded to spray it with a salt water solution regularly over the course of the next day and saw how the salt crystals slowly grew. Having used nylon strings we wanted to test other especially more organic materials such as cotton or wool as well as wick. We then build a smaller cube, with the same concept, which we immersed in a water and salt solution.


Meanwhile we researched the structure of the crystallisation process itself, i.e. which structure would prone crystallisation in contrast to which one wouldn't. Our results showed that triangles where the most suitable structure. In fact triangles that would form themselves by creating a new corner in the center of an already existing one.

Such structures we tried to print in 3D with several materials (wood, sponge like materials and flexible PLA). Unfortunately only the wooden mixture worked out, so we soaked this one in a mixture of salt, sugar and water. In the same solution we added the structure cut out of cardboard, however, both materials did not work well for a crystallisation process due to lack of suction.
We then went back to versions of plexiglas and strings. After finally finding both the wool and the wick we experimented with them and both worked quite well. However, we had to realise that the nylon string, against expectation, worked out the best. Whilst researching chemicals that would crystallise faster than what we had tried until now, we wanted to work with sodium acetate as well as copper sulphate. After numerous calls and visits at even more drugstores and pharmacies, we finally found a small amount of copper sulphate. We had woven a crystallisation structure out of wick and wool and immersed it into the sulphate. After 3 hours we found an astonishing result. We also discovered crystals on the bottom of our plate, which we cultivated further by adding more sulphate and water. Overnight the crystals grew bigger and we found a satisfying outcome. The sulphate being rather difficult to get as well as being rather challenging to handle we decided to keep it as an experiment.
The experiments with the sodium acetate were very quick. The chemical reaction releases energy in form of heat which inspired our further ideas.
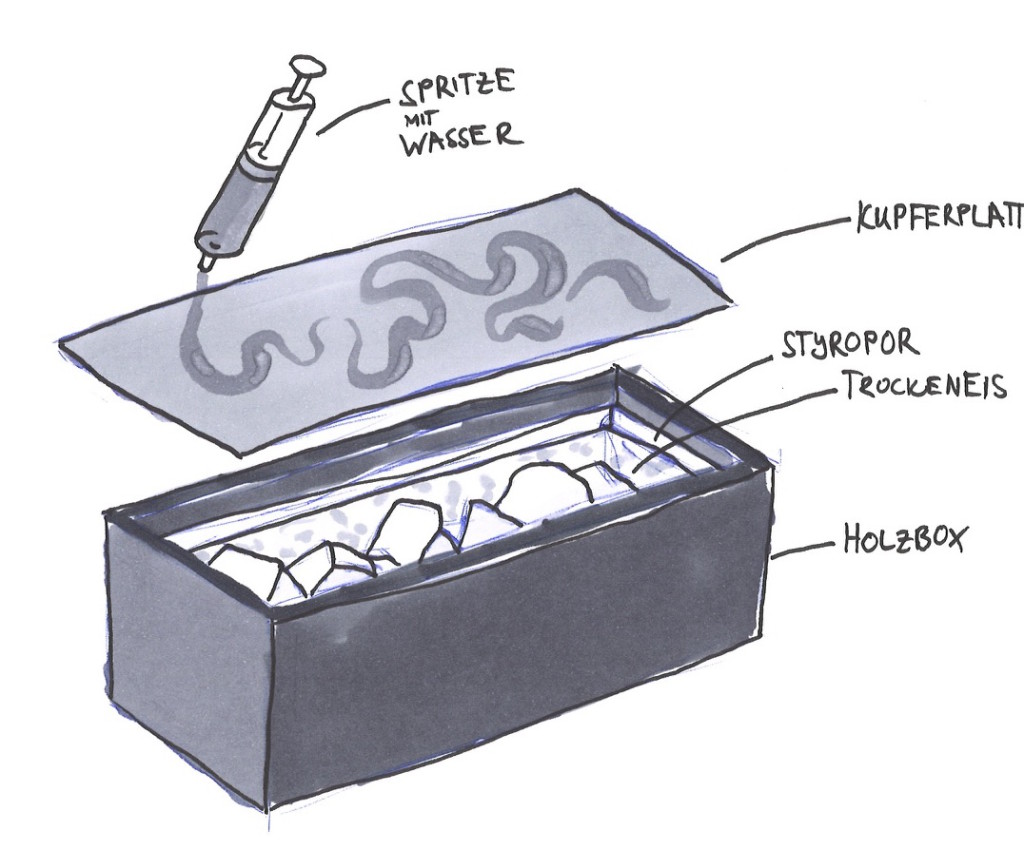
On of our first idea was based on the crystallisation of water through cooling, i.e. ice crystals. We wanted to create a box filled with dry ice and covered by a copper plate, in order to conduct the cold temperature. This box was meant to be a writing area, so that writing with water would directly, with help of the dry ice, produce a crystallised writing. We had tried it with small amounts of dry ice and it worked fine, but since the task was to incorporate human interactions into the process of crystallisation, we decided against this idea, as the interaction seemed to be too distant.
First trials to work with the combination of dry ice, copper, water and a crystallisation structure.
Thinking of the connection of crystallising and eating the idea came up to work with sodium acetate, in order to build a self-heating tupperware. The activation of a trigger point would activate the crystallisation process which again would heat up the inside of a tupperware. Again we discarded this idea, due to the fact that the human interaction was minimal.
Experiments with tupperwares and aluminium foil as well as with sodium acetate itself.
We were still fascinated by the heat produced during the crystallisation process which led us to base our third concept on it as well. The idea was to create a shoe with a sole filled with sodium acetate. The movement and the stepping on the acetate would activate the process by which heat would be conducted. We imagined a suit to go together with the shoe so that we would have a thermal suit activated by the users motion. This idea would incorporate the human interaction but would only have resulted in an additional gadget in everyday life. The course though asked for thoughts outside the box.
The others ideas not being quite satisfying we came up with our final one. Based off the idea of 3D printed prosthesises we first wanted to build a prothesis out of the structure of the crystallisation process. The first issue we had with this ideas, was the fact that even though crystals would grant support they still would be rather edgy and appear aggressive. The second issue we faced was that the process would take a rather long time, which would not be ideal for an artificial limb. The solution we came up with was following:
A flexible and crystallising orthesis made out of smaller plexiglas parts for reinforcement and a woven structure of wick for the flexibility. This particular combination would allow movement while simultaneously provide support and disable not adequate movements. The crystallisation process would occur over time and build up in those ares, which, on one hand needed most support and on the other hand were the most immobile. In this way the orthesis would enable movement by being flexible and not crystallised on the moveable parts of the body, such as junctions. The stability provided by the crystals would therefore be in the less flexible areas.
We realised this idea in form of one part of the full orthesis, which, in lack of time, we could not fully crystallise. Therefore we added a wire frame to stabilise it during the crystallisation process. For presentation purposes we added velcro stripes, which allows to actually try and wear the prototype.
Furthermore we built a second prototype in form of a whole leg prothesis, stabilised only through a frame out of strings and plexiglass. This installation we immersed in water and salt and hung it from the ceiling.
Eventually we hope to have two fully crystallised prototypes and it is only a matter of time until we have a good result.